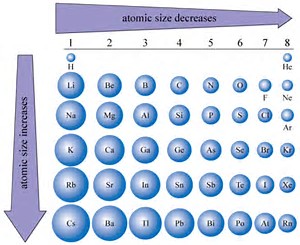
These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Which is a measure of the size of fluorine?Īvailable fluorine properties… More properties… One measure of size is the element-element distance within the element. What is the atomic radius of chlorine and phosphorus?ġ5. It is known that the Van der Waal’s radius is greater than the covalent radius. Which is smaller, Flourine or neon atomic radii?Ġ.72, 1.60 The radius of flourine is measured using covalent radius whereas that of neon is measured using Van der Waal’s radius. What is the atomic radius of a fluorine atom?Ītomic Radius of Fluorine The atomic radius of Fluorine atom is 64pm (covalent radius). But, F has 9 protons attracting those other electrons wheres O has only 8 protons attracting those outer electrons, This will tend to make F a little smaller in radius than O. The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance.īoth F and O are in period 2 meaning they both have 2 shells of electrons. As you can see from the diagrams, the same atom could be found to have a different radius depending on what was around it. The radius of an atom can only be found by measuring the distance between the nuclei of two touching atoms, and then halving that distance. Fluorine has a smaller radius than lithium because of the greater positive charge of the nucleus, pulling electrons and the electron shells in closer.Ĭaesium/Van der Waals radius How do I find the atomic radius? Does F have a small atomic radius? Why does fluorine have the smallest atomic radius?įluorine has more protons resulting in a greater nuclear charge, causing a greater attraction for electrons. What is the radius of a fluorine atom in Metres?īy the definition above, the covalent radius of F is calculated as 71 picometres (1 picometre = 10−12 metres). As atomic number increases, so does the number of positive protons in the nucleus. Why does fluorine have a smaller atomic radius than carbon? Explanation: Atomic radius decreases as you move left to right on the periodic table. It is often denoted by a0 and is approximately 53 pm….Atomic radius. The Bohr radius is consequently known as the “atomic unit of length”. How many atomic radius does fluorine have? 9 Which is a measure of the size of fluorine?.8 Which is smaller, Flourine or neon atomic radii?.7 What is the atomic radius of a fluorine atom?.4 Why does fluorine have the smallest atomic radius?.2 Why is the atomic radius of fluorine?.1 How many atomic radius does fluorine have?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
